Guide quick menu
A practical guide for research institutions, universities, and government-funded organizations that need governed document management across the full research administration lifecycle — from grant application through ethics review, compliance reporting, and long-term research record retention.
Research administration generates a complex, multi-layered document lifecycle that spans grant applications, award documentation, IRB and ethics review records, compliance reporting, research data documentation, and long-term archival obligations. These documents are governed by multiple overlapping frameworks: funding agency requirements, institutional policies, ethics board mandates, and regulatory obligations.
FormKiQ provides a governed document management platform for research institutions that need to manage this complexity within infrastructure they control, with metadata governance, workflow automation, and AI processing capabilities aligned to the specific requirements of research administration programs.
Who It's For
This guide is for research administration offices, grants management teams, sponsored programs offices, IRB and ethics review boards, compliance officers, research deans, CIOs, and IT leaders at universities and research institutions responsible for the document infrastructure supporting research programs. It is also relevant for government agencies managing funded research programs and research-intensive organizations in life sciences, clinical, and applied research sectors.
When to Use It
Use this guide when the volume, complexity, or compliance sensitivity of research administration documents exceeds what can be managed through ad hoc processes, shared drives, or tools designed for other purposes.
- Grant applications, award documentation, and compliance reports are spread across email, shared drives, departmental file servers, and individual researchers' workstations.
- IRB and ethics review records require formal version control, access restrictions, and audit trails that general-purpose file storage cannot provide.
- The institution manages grants from multiple funding agencies, each with distinct documentation, reporting, and retention requirements.
- Compliance reporting is assembled reactively for audits or reviews rather than managed proactively as part of ongoing operations.
- Research data documentation, including data management plans and data use agreements, requires governed management separate from the research data itself.
- Data residency requirements restrict where research records can be stored and processed.
- The institution wants to apply AI processing to research administration documents while keeping AI inside a governed environment with human review, audit trails, and access controls.
- The institution is preparing for a funding agency audit, accreditation review, or regulatory inspection where document governance is itself part of compliance evidence.
The Research Administration Document Landscape
Research administration generates documents across the entire lifecycle of a research program: initial idea, funding application, award, ethics review, active research, compliance reporting, publication, and long-term archival retention. A useful governance model must serve this lifecycle without creating unnecessary administrative burden.
Grant Application and Award Documentation
The grant lifecycle begins with application preparation: project narratives, budgets, biosketches, letters of support, institutional certifications, and supplementary materials. These documents are collaborative, time-sensitive, and often subject to institutional review before submission. After submission, the lifecycle continues through agency correspondence, award notices, negotiation documentation, sub-awards, budget modifications, no-cost extensions, and closeout documentation.
| Document Category | Examples | Governance Considerations |
|---|---|---|
| Application materials | Proposals, budgets, biosketches, letters of support, institutional certifications | Version control during preparation; access limited to project teams and sponsored programs; retention per agency requirements after submission. |
| Award documentation | Award notices, terms and conditions, negotiation correspondence, sub-awards | Controlled access; retention aligned to award period plus agency-specific post-award retention requirements. |
| Financial documentation | Budget justifications, expenditure reports, cost-sharing documentation, effort certifications | Access restricted to financial personnel and PIs; retention aligned to agency and audit requirements. |
| Compliance documentation | Conflict of interest disclosures, export control assessments, biosafety approvals | Sensitive access controls; retention per institutional policy and regulatory requirements. |
| Progress and closeout | Annual reports, final reports, invention disclosures, equipment disposition | Version control for drafts; retention per agency closeout requirements. |
IRB and Ethics Review Records
IRB and ethics review records document institutional oversight of research involving human subjects: protocols, informed consent forms, adverse event reports, continuing review documentation, committee deliberations, and decisions. These records require formal version control, strict access controls, and long-term retention.
| IRB Document Type | Governance Requirements |
|---|---|
| Research protocols | Version control with full history; ability to reconstruct which version was in effect at any point; access restricted to PI, study team, IRB staff, and authorized reviewers. |
| Informed consent forms | Version-controlled with approval dates; traceable to the IRB approval authorizing each version; retention per regulation and institutional policy. |
| Adverse event reports | Timely documentation with timestamps; access controlled; notification workflows to IRB and regulatory bodies. |
| Continuing review documentation | Periodic review submissions with approval records, scheduling, and deadline tracking. |
| IRB meeting minutes and decisions | Restricted access; formal retention requirements; documentation of the basis for each IRB determination. |
| Correspondence | Communications between IRB and investigators retained alongside the study record. |
Compliance Reporting
Research compliance reporting spans federal regulations, funding agency terms and conditions, and institutional policies. Compliance documentation is often assembled from departmental records, financial systems, training databases, IRB records, and investigator-maintained files. The challenge is not just producing documentation, but demonstrating that it is complete, current, and governed throughout its lifecycle.
Research Data Documentation
Research data documentation is distinct from the research data itself. It includes data management plans, data sharing agreements, de-identification records, data use agreements, data security plans, and documentation of data handling procedures. FormKiQ manages the documentation layer: the plan, agreement, approval, and evidence that describe how research data is governed.
Metadata Governance for Research Documents
Research administration documents require rich, structured metadata to be findable, governable, and compliant. FormKiQ's configurable metadata schemas support classification by multiple dimensions at the same time.
| Metadata Dimension | Purpose | Examples |
|---|---|---|
| Project or grant identifier | Links documents to specific research projects or awards | NIH grant number, institutional project ID, sponsor reference number |
| Funding source | Identifies the funding agency and program | NIH/NIGMS, NSF, CIHR, Wellcome Trust, institutional internal funding |
| Principal investigator | Associates documents with the responsible researcher | PI name, institutional identifier, department |
| Document type | Classifies the document within the research administration taxonomy | Protocol, consent form, progress report, expenditure report, IRB decision letter |
| Compliance status | Tracks the document's compliance state | Pending review, approved, expired, requires renewal, under audit |
| Reporting period | Links documents to reporting cycles | Annual report period, project year, fiscal year |
| Regulatory framework | Associates documents with applicable regulations | Common Rule, HIPAA, Export Control, FCOI |
| Retention category | Determines retention schedule and disposition rules | Award records, IRB records, financial records, clinical trial records |
Workflow Automation for Research Administration
Research administration workflows are multi-step, multi-stakeholder, and often deadline-driven. FormKiQ supports routing, review, approval, notification, and escalation patterns common to research programs.
Grant application review and institutional approval
Route proposals through departmental review, sponsored programs review, institutional sign-off, and submission preparation with deadline tracking and escalation.
IRB protocol review
Support submission intake, completeness checking, reviewer assignment, committee preparation, decision documentation, and investigator notifications.
Compliance reporting
Trigger scheduled reporting workflows, collect documentation from multiple sources, route reports for review and approval, and track submission status.
Award modifications
Route budget modification requests, no-cost extension requests, and scope change requests through the appropriate research, financial, and institutional channels.
Workflows produce audit trails that document who submitted, who reviewed, what decision was made, when it happened, and what happened next.
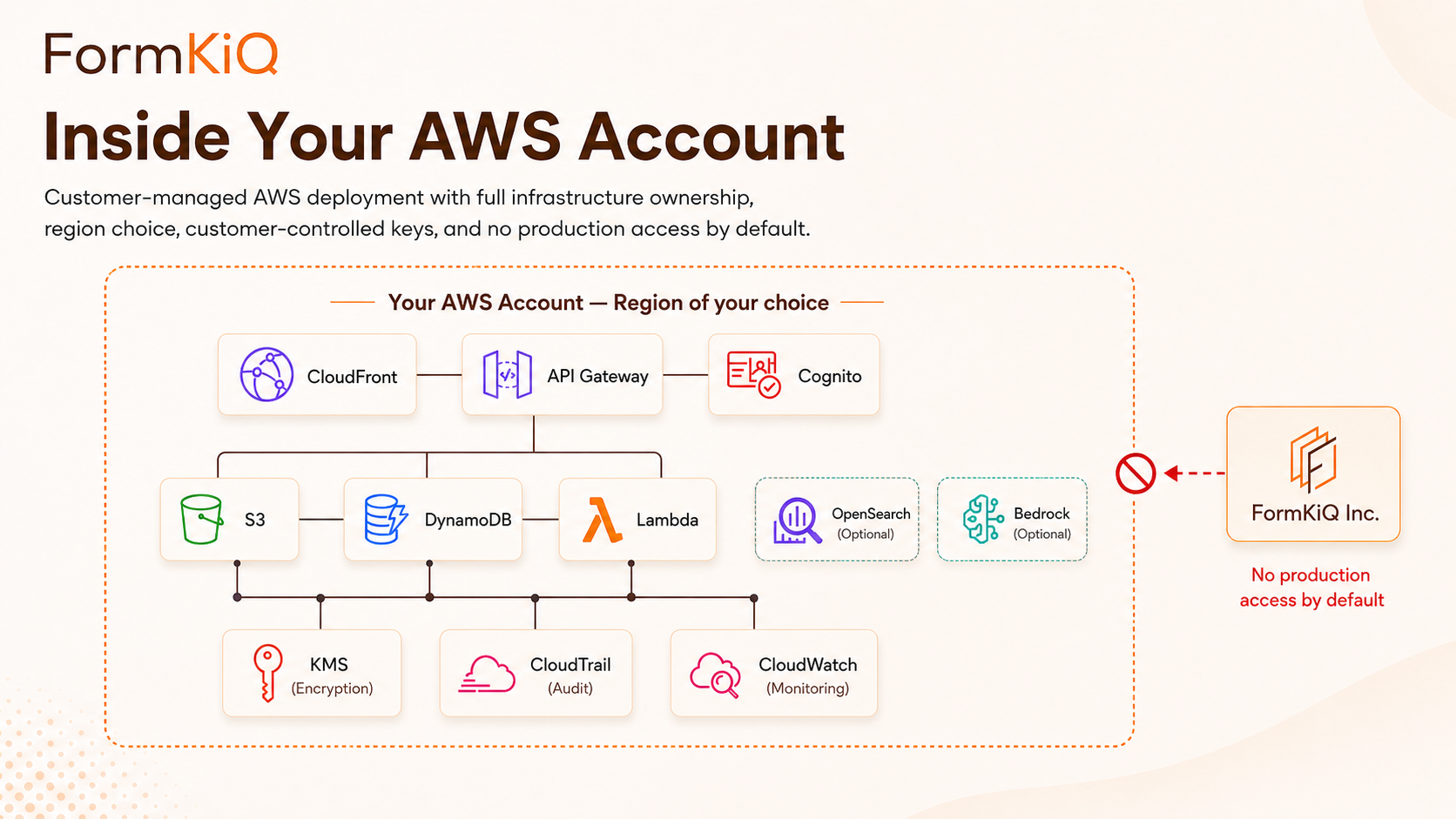
Multi-Tenant Architecture for Research Institutions
Large research institutions often manage research administration documents across faculties, departments, research centers, institutes, affiliated hospitals, and administrative units. Each may have its own compliance needs and access-control boundaries.
FormKiQ's multi-tenant architecture supports this through sites: logically isolated document environments within a single deployment. Each site can have its own document types, metadata schemas, workflows, access controls, and retention policies while sharing the same platform infrastructure. For multi-institutional research programs and consortia, separate FormKiQ deployments in separate AWS accounts can provide institutional boundary isolation while supporting controlled cross-institution document sharing.
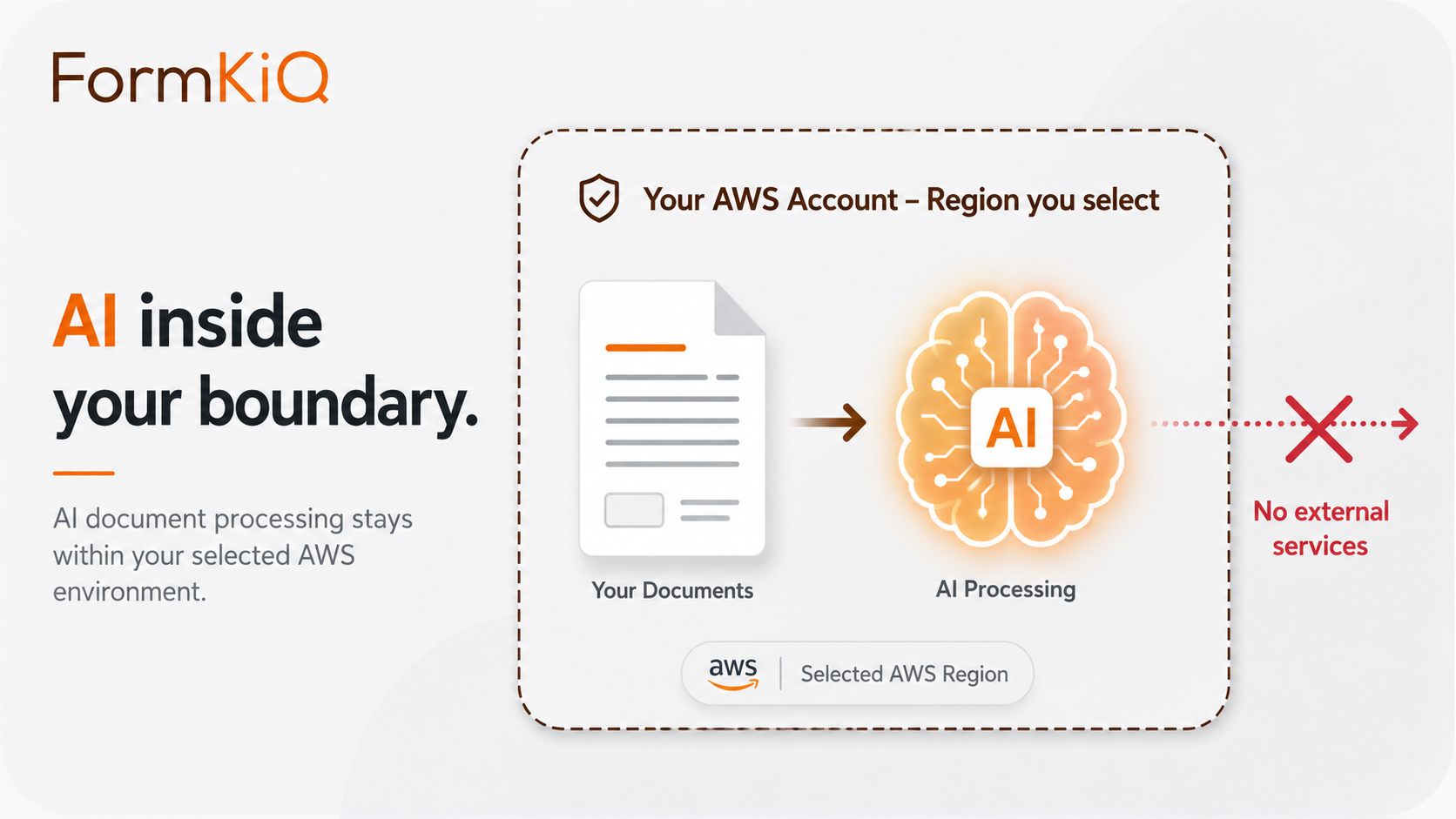
AI Processing for Research Administration
AI processing through Amazon Bedrock can accelerate research administration workflows, but research compliance decisions still require human review. FormKiQ treats AI outputs as governed, reviewable metadata and workflow signals.
| AI Capability | Research Administration Application | Governance Consideration |
|---|---|---|
| Document classification | Identify proposals, budgets, consent forms, progress reports, expenditure reports, and decision letters. | Classification results are reviewable, correctable, auditable metadata. |
| Metadata extraction | Extract grant numbers, PI names, funding sources, dates, amounts, study identifiers, and reporting periods. | Extracted values can route to human review before becoming authoritative metadata. |
| Compliance analysis | Screen submitted protocols or reports against compliance checklists for missing elements, incomplete disclosures, or potential issues. | AI analysis is a screening aid, not a compliance determination. |
| Summarization | Summarize lengthy proposals, reports, or correspondence for reviewer triage and committee preparation. | Summaries inherit document access controls. |
| Gap identification | Analyze document collections for missing required documents in a grant, study, or compliance package. | Gap analysis results are workflow signals that trigger follow-up, not automated compliance actions. |
Retention and Archival for Research Records
Research record retention requirements vary by document type, funding agency, regulatory framework, and institutional policy. FormKiQ supports configurable retention schedules at the document type, project, or individual document level.
| Retention Category | Typical Requirement | Source |
|---|---|---|
| Federal grant records | Often 3 years after submission of final expenditure report | 2 CFR 200.334 and agency-specific terms |
| IRB records | Duration of study plus additional years depending on regulation and institution | 45 CFR 46 and institutional policy |
| Clinical trial records | Often 15 years or longer depending on jurisdiction and trial type | FDA 21 CFR 11, ICH E6, institutional policy |
| Financial records | Often 3-7 years after closeout depending on agency and audit requirements | Agency-specific terms and conditions |
| Export control records | Often 5 years after expiration of license or completion of transaction | EAR/ITAR requirements |
| Intellectual property records | Life of patent plus institutional policy period | Institutional IP policy |
Data Residency for Research Institutions
Research institutions in Canada, the EU, Australia, and other jurisdictions may have data residency requirements that specify where research records must be stored and processed. This can be especially important for records containing personal information, participant documentation, clinical research materials, or unpublished research findings.
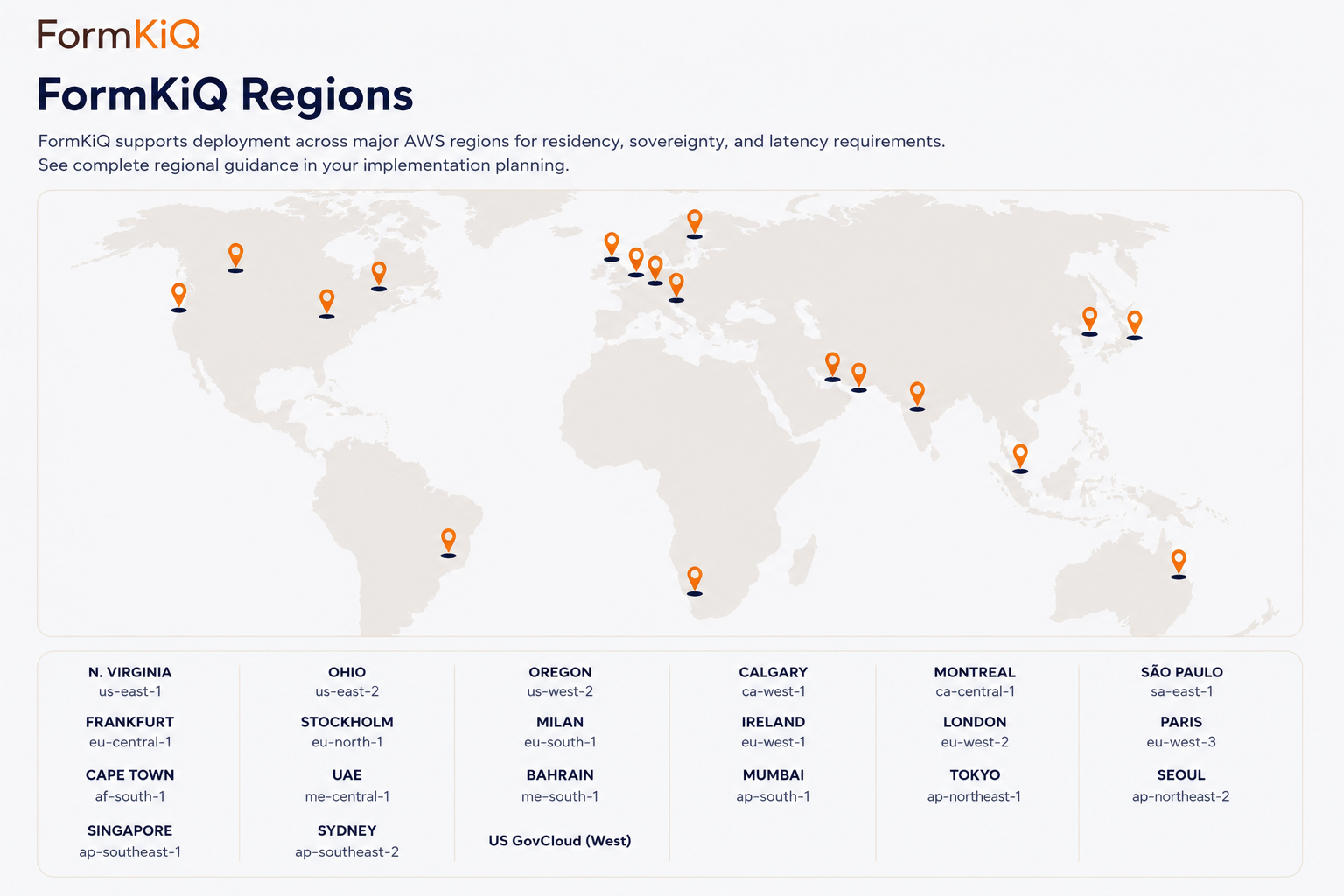
FormKiQ deploys into supported AWS regions so institutions can align document storage and AI processing with residency programs. Canadian institutions can deploy in Canadian AWS regions, EU institutions can deploy in EU regions, and Australian institutions can deploy in Sydney, subject to AWS service availability and institutional architecture decisions.
Integration with Research Administration Systems
Research institutions typically operate grants management systems, financial systems, IRB management systems, and compliance training systems. FormKiQ is not a replacement for those systems; it is the governed document layer that connects to them.
Grants systems
Cayuse, Kuali, InfoEd, ERA Commons, and other systems can submit or retrieve award documentation through API patterns.
Financial systems
Workday, PeopleSoft, Banner, and ERP systems can connect expenditure documentation and financial records to governed retention.
IRB systems
IRBManager, Advarra, RAMP, and similar platforms can store and retrieve protocols, decisions, and correspondence.
Training systems
Compliance training systems can deposit completion certificates, acknowledgments, and related compliance evidence.
Recommended Platform Configuration
Edition
FormKiQ Advanced is the recommended starting point for research institutions. It provides document management, workflows, ABAC access controls, encryption, AI processing through Amazon Bedrock, enhanced full-text search, and multi-instance licensing.
Enterprise Fit
FormKiQ Enterprise is appropriate for large research universities with vendor-managed or hybrid deployment requirements, multi-region operations, custom SLAs, or compliance consulting needs.
| Module | Research Administration Use |
|---|---|
| Enhanced Full-Text Search | Discovery across grant portfolios, IRB records, compliance evidence, and long-term research record collections. |
| KnowledgeBase | Research document discovery and institutional knowledge access across governed collections. |
| AI Processing and Analysis | Classification, metadata extraction, compliance screening, summarization, and gap identification through Amazon Bedrock. |
| Integration Frameworks | Connections to grants management, IRB, financial, HR, compliance training, and institutional systems. |
Getting Started
The evaluation process for research institution deployments starts with a conversation about the institution's research administration landscape: the grant portfolio, regulatory frameworks in play, systems currently in use, current document management pain points, and near-term priorities.
Common starting points include centralizing grant documentation for a specific faculty or department, establishing governed IRB record management, automating compliance reporting workflows, or building a research data documentation program.
Important Guardrails
FormKiQ provides architectural controls that research administration document programs require: access control, audit trails, encryption, retention, workflow automation, and governed AI processing. It does not claim compliance with any specific regulatory framework by default.
Whether a given FormKiQ deployment satisfies the requirements of the Common Rule, HIPAA, PIPEDA, GDPR, or any funding agency's terms and conditions depends on how it is configured, operated, and validated by the institution's compliance, legal, and security teams.
AI processing through Amazon Bedrock produces probabilistic outputs. For research administration, where compliance determinations, IRB decisions, and regulatory reporting have institutional and legal significance, AI outputs are screening aids and workflow accelerators, not authoritative compliance determinations. Human review is a required component of governed AI processing for research administration applications.
Related Links
Business Solutions · Research and Grant Administration · AI Processing and Analysis · Deployment & Compliance · Data Residency







