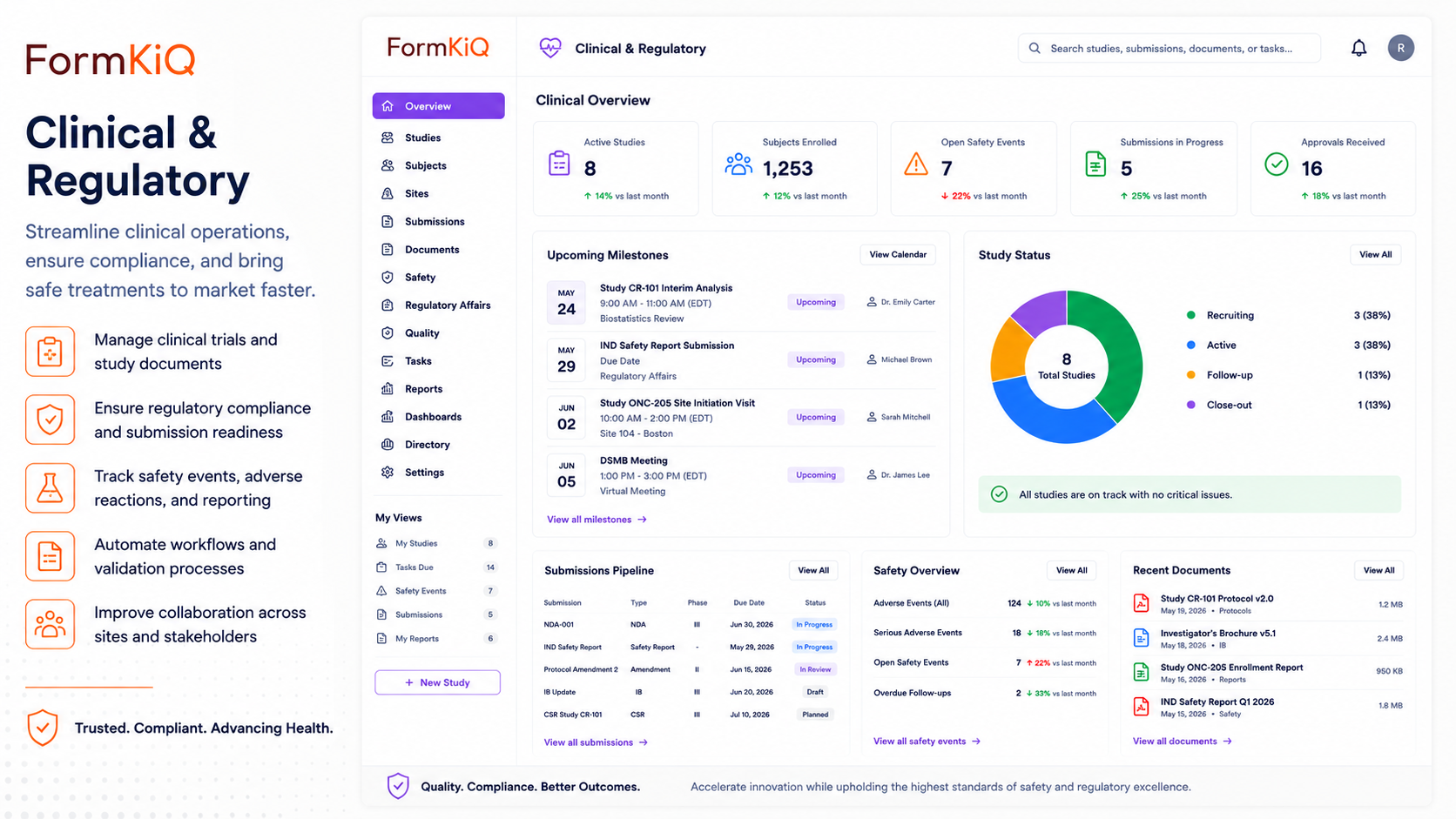
FormKiQ's Clinical and Regulatory Document Management solution provides a governed document layer for organizations operating under FDA, EMA, ICH, and similar regulatory frameworks — where document control is not an operational preference but a regulatory requirement.
What Clinical and Regulatory Document Management covers
Manage clinical trial documentation, regulatory submissions, quality system records, and supporting compliance documentation with the version control, access management, audit trails, and retention policies that life sciences regulatory frameworks require.
- Controlled document authoring, review, approval, and distribution workflows aligned to quality management system requirements
- Version control and change management that ensures document histories are complete, traceable, and accessible without confusion with current controlled documents
- Access management enforced at the document level with role-based permissions appropriate to regulatory environments
- Audit trails for every document action — creation, review, approval, distribution, and access
- Retention policies configured to regulatory framework requirements by document type and jurisdiction
- Regional deployment options to support multi-jurisdiction regulatory submission requirements
Regulatory document model
The guide focuses on controlled clinical, quality, and regulatory records where authoring, version history, approvals, signatures, submission readiness, and retention evidence must be managed rigorously.
Controlled document set
Manage protocols, SOPs, validation records, regulatory submissions, clinical trial documents, quality records, and supporting evidence.
Regulatory metadata
Track study, product, site, region, document type, lifecycle state, approval status, version, owner, and retention requirements.
Version and approval control
Preserve draft, review, approved, superseded, and archived states with a complete audit trail.
Submission support
Organize final approved documents and supporting materials for regulatory packages and inspection readiness.
Regulatory document lifecycle
FormKiQ supports controlled authoring and regulated document workflows from creation to archive.
| Stage | How FormKiQ Supports It |
|---|---|
| Authoring | Create or ingest controlled documents with metadata, ownership, and document type classification. |
| Review | Route drafts through scientific, clinical, regulatory, quality, or legal review. |
| Approval and signature | Record approvals and electronic signature evidence where required. |
| Distribution and submission | Publish approved versions, assemble submission packages, and manage controlled access. |
| Retention and archive | Retain superseded and final versions according to regulatory schedules. |
Automation and governance focus
AI-assisted analysis
Extract product names, study identifiers, sites, dates, regulatory references, and summary data from controlled documents.
Controlled authoring
Support check-in/check-out, version comparison, approval routing, and change history.
Enterprise integration
Connect regulatory records with QMS, CTMS, RIMS, ERP, MES, and training management systems.
Recommended platform configuration
Edition
FormKiQ Enterprise
Modules commonly used
Document Generation, Enhanced Full-Text Search, AI Processing and Analysis
Common fits: pharmaceutical and biopharmaceutical clinical and regulatory documentation, medical device quality management system documentation, contract research organization document management, and any life sciences organization with formal document control obligations under applicable regulatory frameworks.
Read the in-depth guide
For a deeper walkthrough of regulatory document lifecycle, AI analysis, controlled authoring, electronic signatures, submission assembly, metadata search, compliance alignment, and AWS deployment models, read the full guide.